Targeting epiregulin to the treatment-damaged tumor microenvironment curbs therapeutic resistance
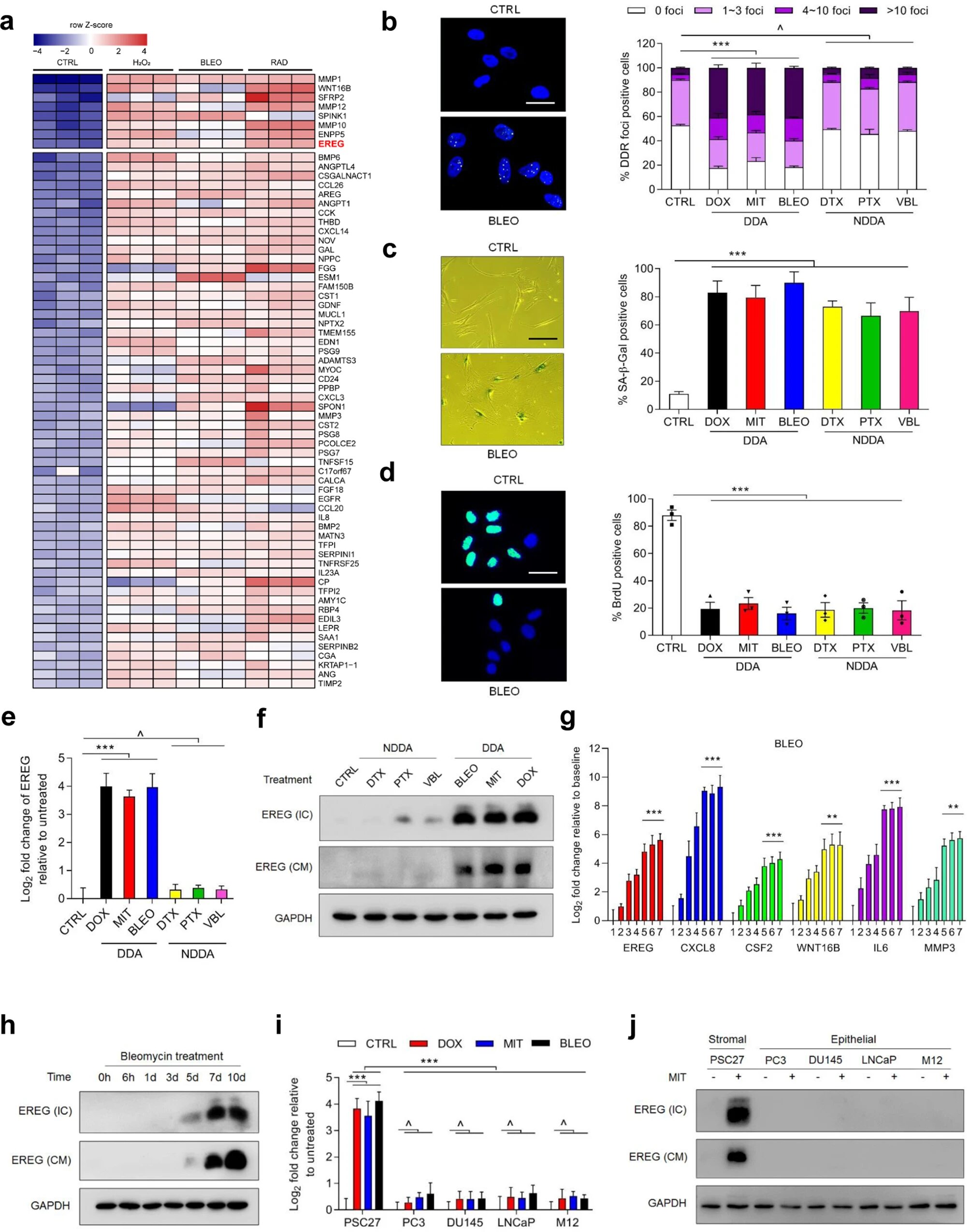
Genotoxicity induces the expression of EREG and other secreted factors of the SASP spectrum in human stromal cells. a Transcriptome profiling of gene expression changes in a primary normal human prostate stromal cell line (PSC27) using microarrays. Cell lysates were collected for analysis 7 days after treatment. CTRL control. H2O2 hydrogen peroxide. BLEO bleomycin. RAD radiation. Red, EREG. Agilent microarray data adapted from Sun et al. with permission from Natural medicinecopyright 2012, Springer Nature. b Representative images of immunofluorescence staining (co-staining of γH2AX and p-53BP1, left) and comparative statistics (right) of DNA damage response (DDR) in PSC27 cells treated with DOX (doxorubicin), MIT (mitoxantrone), BLEO (bleomycin), DTX ( docetaxel), PTX (paclitaxel) and VBL (vinblastine). DDA DNA-damaging agents (DDA). NDDAs are non-DNA damaging agents. DDR were classified into four subcategories including 0 foci, 1–3 foci, 4–10 foci, and >10 foci per cell. Scale bars, 15 μm. c SA-β-Gal staining of PSC27 cells treated with different agents used in b. Cells were stained 7 days after in vitro treatment. Scale bars, 30 μm. That’s right, comparative statistics. d BrdU staining of stromal cells treated with different agents as indicated in b and c. Scale bars, 15 μm. That’s right, comparative statistics. d Quantitative RT-PCR of EREG expression after treatment of PSC27 cells with different agents. Cell lysates were collected for measurement 7 days after treatment. Signals normalized to CTRL. e Immunoblot analysis of EREG expression in stromal cells 7 days after treatment performed as directed. IC of intracellular samples. CM conditioned media. GAPDH, loading control. Mr Time-course expression assessment of a subset of EREG and other typical SASP factors (CXCL8, CSF2, WNT16B, IL6 and MMP3) after in vitro drug treatment of stromal cells. Numerals indicate individual days after treatment. h Immunoblot measurement of EREG expression at the protein level over time as described in Mr. i Comparative evaluation of EREG transcript expression in stromal cells (PSC27) versus cancerous epithelial cells (PC3, DU145, LNCaP and M12). Signals normalized to untreated sample per cell line. j Evaluation of EREG expression in protein lysates of stromal and epithelial cells after bleomycin treatment by immunoblot performed in i. Data are representative of three independent experiments. ^old > 0.05, *old < 0.05, **old < 0.01, ***old < 0.001. old values were calculated by the Student ttest (c–d, Mr) and two-way ANOVA (b, i). ^old> 0.05, *old< 0.05, **old< 0.01, ***old< 0.001. credit: Oncogene(2022). DOI: 10.1038/s41388-022-02476-7
A recent study published in Oncogeneand led by Dr. Sun Yu’s group from the Shanghai Institute of Nutrition and Health (SINH) of the Chinese Academy of Sciences discovered the role and functional mechanisms of senescence-related secretory factor epiregulin (EREG) in the treatment-damaged tumor microenvironment, providing a novel therapeutic target and non-invasive biomarker for malignancy human tumors including prostate cancer.
Cellular senescence is a unique cellular state with many distinct and unique features. Among these cell characteristics, the unique secretory phenotype of senescent cells, namely the senescence-associated secretory phenotype (SASP), enables the complex function of signaling to other cells in the microenvironment.
This secretory phenotype has been shown to contribute significantly to disease progression in the elderly, including chronic inflammatory processes. Therefore, the study of SASP-related factors and their functions in tumor and other pathological microenvironments has far-reaching implications for the clinical treatment of geriatric diseases.
In this study, the researchers confirmed that EREG expression is significantly increased under senescence-induced conditions of DNA damage by senescence induction in primary human stromal cells. Analysis of cancer samples from clinical prostate cancer and breast cancer patients before and after chemotherapy showed that EREG is significantly upregulated in senescent paracancerous stromal cells.
Mechanistically, DNA damage induces nuclear localization of the transcription factor NF-κB in stromal cells and binds to multiple sites in the EREG promoter region, thereby contributing to the regulation of EREG expression after cellular senescence. Other aging-related factors have also been reported to promote EREG transcription. In the tumor microenvironment, EREG activates multiple signaling pathways by binding to EGFR receptors on the surface of adjacent cancer cells and causes exacerbation of malignant phenotypes, such as cancer cell proliferation, migration and invasion, and emergence of tumor drug resistance.
Using RNA-Seq analysis, the researchers identified a ubiquitin ligase, MARCHF4, which was significantly upregulated in EREG-stimulated cancer cells. MARCHF4 can down-regulate E-cadherin expression in cancer cells and inhibit cancer cell apoptosis, leading to drug resistance.
U mouse model, the combination of an EREG monoclonal antibody and an EGFR monoclonal antibody significantly reduced tumor volume and significantly prolonged the progression-free period. It should be noted that the expression level of EREG in clinical cancer patients can effectively and accurately predict the effectiveness of cancer treatment.
This study elucidated the functions and regulatory mechanisms of the novel senescence-associated secretory factor EREG in tumor microenvironment and discovered its importance in translational medicine.
Changxu Wang et al. Targeting epiregulin in the treatment-damaged tumor microenvironment curbs therapeutic resistance, Oncogene(2022). DOI: 10.1038/s41388-022-02476-7
Provided
Chinese Academy of Sciences
Citation: Targeting epiregulin in the treatment-damaged tumor microenvironment curbs therapeutic resistance (2022, October 25) Retrieved October 25, 2022 from https://medicalxpress.com/news/2022-10-epiregulin-treatment-damaged-tumor -microenvironment-restrains.html
This document is subject to copyright. Except in good faith for the purpose of private study or research, no part may be reproduced without written permission. The content is provided for informational purposes only.


